Sangamo Gains FDA Clearance To Begin In-Vitro Gene Editing Treatment For Hemophelia
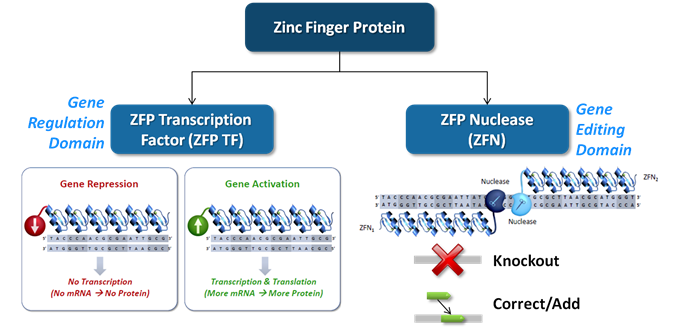
Sangamo, a leader in therapeutic gene editing, announced that the FDA has cleared the company to begin an in-vitro gene editing treatment therapy for hemophelia. They are the first company to be cleared for human gene editing in the United States and currently the only company to be doing so on an embryonic level. The gene editing treatment is based on Sangamo’s proprietary In Vivo Protein Replacement Platform™ ( IVPRP ™), and will now allow the company to begin investigating the efficacy of this therapy for treating hemophilia.
This is going to usher in a new era of genetic medicine and hopefully this gene editing initiative will herald in further genetic editing programs in addition to expanding genomic health projects.
Sangamo’s IVRP gene editing approach works by focusing on the albumin gene locus, a highly expressive and liver specific genomic site that can be worked with safely because it is a phenomenal target for therapeutic gene expression. The gene locus is then altered to permanently produce corrective levels of a protein that address enzymatic shortcomings common to hemophilia. This treatment method can also be applied to the treatment of lysomal storage disorders. It is believed that this method of treating hemophilia will be more effective than previous gene therapies such as AAV which were rendered ineffective when liver cells in pediatric patients began to replicate and grow.
Gene Editing To Replace Protein Deficiencies
While this gene editing treatment is very specific to hemophiliacs and other lysomal storage disorders, this news is tremendous for healthcare but specifically genetic medicine. With many rare genetic diseases affecting patients worldwide, whom previously had little to no hope for much more than simple maintenance of their symptoms there now may in fact be genetic cures to their diseases. While there will always continue to be diseases and ailments that are beyond the scope of modern medicine to some extent with genetic medicine becoming more accessible and applied in a real world situation, we could see over the next decade an abundance of previously hard to treat conditions become fairly simple to correct.
 For further information about Sangamo’s new gene editing program or to further explore their gene editing press release
For further information about Sangamo’s new gene editing program or to further explore their gene editing press release
